|
3/1/2023 0 Comments Molar mass of ethanol  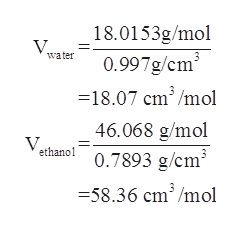
of moles), the mass of 0.5623mol of ethanol is approximately 25.9g. You can see how this formula works reading the example above or/and choosing another one at the bottom of this page. Using the formula mass (molar mass) x (no. That is exactly the formula that we use in this calculator. M kg v m³ = m lb v gal × mcf vcf, but m kg v m³ is, by definition, the density (d) in kg/m³, soįinally, we can generalize this formula any units of mass and volume, since we know how to find the values for mcf and vcf (Check the factor tables below). Now, dividing the equation (1) by the equation (2), we get In Imperial or US customary measurement system, the density is equal to 49.2744 pound per cubic foot lb. density of ethanol is equal to 789.3 kg/m at 20C (68F or 293.15K) at standard atmospheric pressure. Ethanol weighs 0.7893 gram per cubic centimeter or 789.3 kilogram per cubic meter, i.e. Volume in cubic meters = volume in gallons × vcf or Equivalent molar concentration per liter. We will call 0.003785411784 the volume conversion factor - vcf To transform the volume from gallons to cubic meters we should multiply the volume in cubic meters by 0.003785411784. Molecular weight is actually an older term of relative molar mass or molecular mass, which is a dimensionless quantity equal to the molar mass divided by the molar mass constant defined by 1 g/mol. Mass in kilograms = mass in pounds × mcf or The exact term of the above molecular weight is molar mass, which is based on the atomic mass of each element. We will call 0.45359237 the mass conversion factor - mcf 
To transform the mass from pounds to kilograms we should multiply the mass in pounds by 0.45359237. In addition alcohol have a strong C-O stretch near 1000 cm-1. If we have, for example, mass in pounds and volume in gallons, we should do something to correct our result. The mass spec of D provides a molecular formula of C 5 H 10 O 2. For example, mass in kg and volume in liters. But this works fine when the density value we have (from a chart for example) is defined using the same unit of volume and mass that we will use in the formula. Very simple isn't it? Yes, it is as simple as that. Mass = density × volume = 750 × 2 = 1500 Kg Plugging the values in the formula, we get Well, by definition of density we just need to use the formula:Įxample: How much does 2 cubic meters of gasoline weigh in kilograms if the gasoline density is 750 kg/m³? How to convert from volume to mass (incorrectly called weight)? Convert grams CH3CH2OH to moles or moles CH3CH2OH to grams. Volume to Mass Converter (Construction).Weight (Mass) to Volume to Converter for Recipes.Volume to (Weight) Mass Converter for Recipes.In mass spectrometry, the molecular mass of small molecules (less than about 200 atoms of a. Please let us know how we can improve this web app. Soluble in cold water, ethanol and methanol. Related: Molecular weights of amino acids Weights of atoms and isotopes are from NIST article. Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations:ĭefinitions of molecular mass, molecular weight, molar mass and molar weight Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Ts, Tos, Bz, TMS, tBu, Bzl, Bn, Dmg.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Computing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
